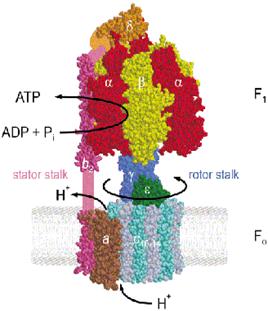
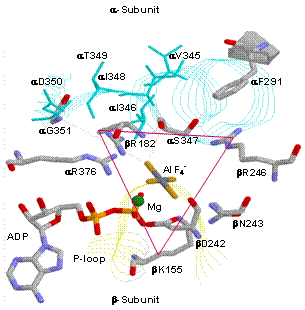
|
Publications
Ahmad, Z. and Laughlin, T. F. (2010) Medicinal Chemistry of ATP synthase: a
potential drug target of dietary polyphenols and amphibian antimicrobial
peptides, Current Medicinal Chemistry (In Press).
Chinnam, N., Dadi, P.K., Sabri, S.A., Ahmad, M., and Ahmad, Z. (2010)
Dietary bioflavonoids inhibit Escherichia coli ATP synthase in a differential
manner Int. J. Biol. Macromol. 46, 478-486.
Laughlin, F. and Ahmad, Z. (2010) Inhibition of Escherichia coli
ATP synthase by amphibian antimicrobial peptides. Int. J. Biol. Macromol
46, 367-374.
Dadi, P. K.,
Ahmad, M., and Ahmad, Z. (2009) Inhibition of ATPase activity of Escherichia
coli ATP synthase by polyphenols Int. J. Biol. Macromol. 45, 72-79.
Li, W., Brudecki,
L.E., Senior, A.E., and Ahmad, Z. (2009) Role of
a-subunit VISIT-DG sequence residues
Ser-347 and Gly-351 in the catalytic sites of Escherichia coli ATP
synthase. J. Biol. Chem. 284, 10747-10754.
Ahmad,
S. and Ahmad, Z. (2008) “ ATP-binding site as a further application of
neural network to residue level prediction” In Proceedings of International
Joint Conference on Neural Networks (IJCNN), World Conference on Computational
Intelligence (WCCI), June 1-8, Hong Kong, IEEE. pp. 2431-2435.
Brudecki LE, Grindstaff JJ & Ahmad Z. (2008) Role of
aPhe-291 residue in the
phosphate-binding subdomain of catalytic sites of Escherichia coli ATP
Synthase Arch. Biochem. Biophys. 471, 168-175.
Ahmad Z. & Senior AE
(2006) Inhibition of the ATPase activity of
Escherichia coli ATP synthase by magnesium fluoride. FEBS Lett.
580, 517-520.
Ahmad Z.
& Senior AE
(2005) Identification of phosphate
binding residues of Escherichia coli ATP synthase. J. Bioenerg.
Biomembr. 37,
437-440.
Ahmad Z.
& Senior AE
(2005) Modulation of charge in the
phosphate binding site of Escherichia coli ATP synthase. J. Biol.
Chem. 280,
27981-27989.
Ahmad Z.
& Senior AE
(2005) Involvement of ATP synthase
residues αArg-376, ßArg-182, and ßLys-155 in Pi binding. FEBS Lett.
579,
523-528.
Ahmad Z.
& Senior AE
(2004) Role of residue ß-Asn-243 in
the phosphate-binding subdomain of catalytic sites of Escherichia coli F1-ATPase.
J. Biol. Chem. 279,
46057-46064.
Ahmad Z & Senior AE (2004) Mutagenesis of residue ß-Arg-246 in the
phosphate-binding subdomain of catalytic sites of Escherichia coli F1-ATPase.
J. Biol. Chem. 279,
31505-31513.
Ahmad A,
Ahmad Z and MA Baig (2003) Hepatic
Sulfite oxidase: effect of anions on its activity. Trends Clinical. Biochem.
Lab Medicine. 1, 751-755.
Ahmad Z, Salim M & Maines MD (2002) Human biliverdin reductase is a leucine
zipper like DNA-binding protein and functions in transcriptional activation of
heme oxygenase-1 by oxidative stress J. Biol. Chem.
277,
9226-9232.
Ahmad Z & Sherman F (2001) Role of Arg-166 in yeast cytochrome C1
J. Biol. Chem. 276,
18450-18456.
Ahmad Z, Yadav S, Ahmad F & Khan NZ (1996) Effects of salts of alkali earth
metals and calcium chloride on the stability of cytochrome c and myoglobin.
Biochem. Biophys. Acta 1294,
63-71.
Ahmad Z
& Ahmad F (1994) Physicochemical characterization of products of unfolding of
cytochrome c by calcium chloride. Biochem. Biophys. Acta. 1207,
223-230.
Ahmad Z & Ahmad F (1992) Mechanism of denaturation of cytochrome c by lithium
salts. Ind. J. Chem. 31b,
874-879.
Research
opportunities
Research
opportunities
for graduates, and undergraduates are available to
work on recently funded NIH research grant. Those of you
who would like to be a part of my research group or would like to learn more
about the the role of ATP synthase in human health and diseases please visit our lab, or email
us at: ahmadz@etsu.edu
Current Lab Members:
Mubeen Ahmad,
Mubeen is lab Technician. She
helps in maintaining day-to-day activities of the lab including, ordering
supplies, helping in dish washing, maintaining E. coli stock cultures,
and preparing media plates. Mubeen also performs and helps in routine
experiments such as biochemical assays, site directed mutagenesis, cloning, and
protein/membrane purification.
Graduate
students:
Laura E. Brudecki,
Laura became a graduate student in the spring of
2009. She initially joined my lab as an undergraduate in the fall 2006. She has
been working on the characterization of Pi binging site residues of E. coli
ATP synthase. Her current work involves the characterization of the
a-subunit
VISIT-DG sequence residues in the Pi binding subdomain of the catalytic sites.
Nagababu Chinnam,
NBC
joined our lab in January 2009 as a graduate student. His is
joining the project with PKD on the inhibitory effects of polyphenols/bioflavonoids.
Sneha Reddy, Sneha
joined our lab in January 2009 as a graduate student. She has joined the
project on characterization of the
a-subunit
VISITDG sequence residues in the Pi binding subdomain of the catalytic sites and
will specifically focus on the DG residues.
Chao Zhao ,
Chao joined us in August 2008. He will be working on
characterization of the
a-subunit
VISITDG sequence residues in the Pi binding subdomain of the catalytic sites and
will focus on the
aI346
and
aI348
residues.
Junior Tayou, Junior also joined
us in August 2008. He is working on
the inhibitory effects of peptides and their binding site on ATP synthse.
Undergraduate students:
Steven Sherman, Nitin Sachan, Saurabh Sachan, Mai Xiong, Shabaaz Sabri, and
Mohammad Bowers
Each of
these students is undertaking their own specific project or has been coupled
with a graduate student.
Teaching
Biochemistry of macromolecules Lecture and lab; Biochemistry of Metabolism lab;
Honors Discussion group; Non-major Biology I, Graduate topics course
|